Cochrane Meta-Analysis
Aprepitant Is the Most Efficacious Compound for the Prevention of Vomiting After Surgery With High-Confidence Evidence1
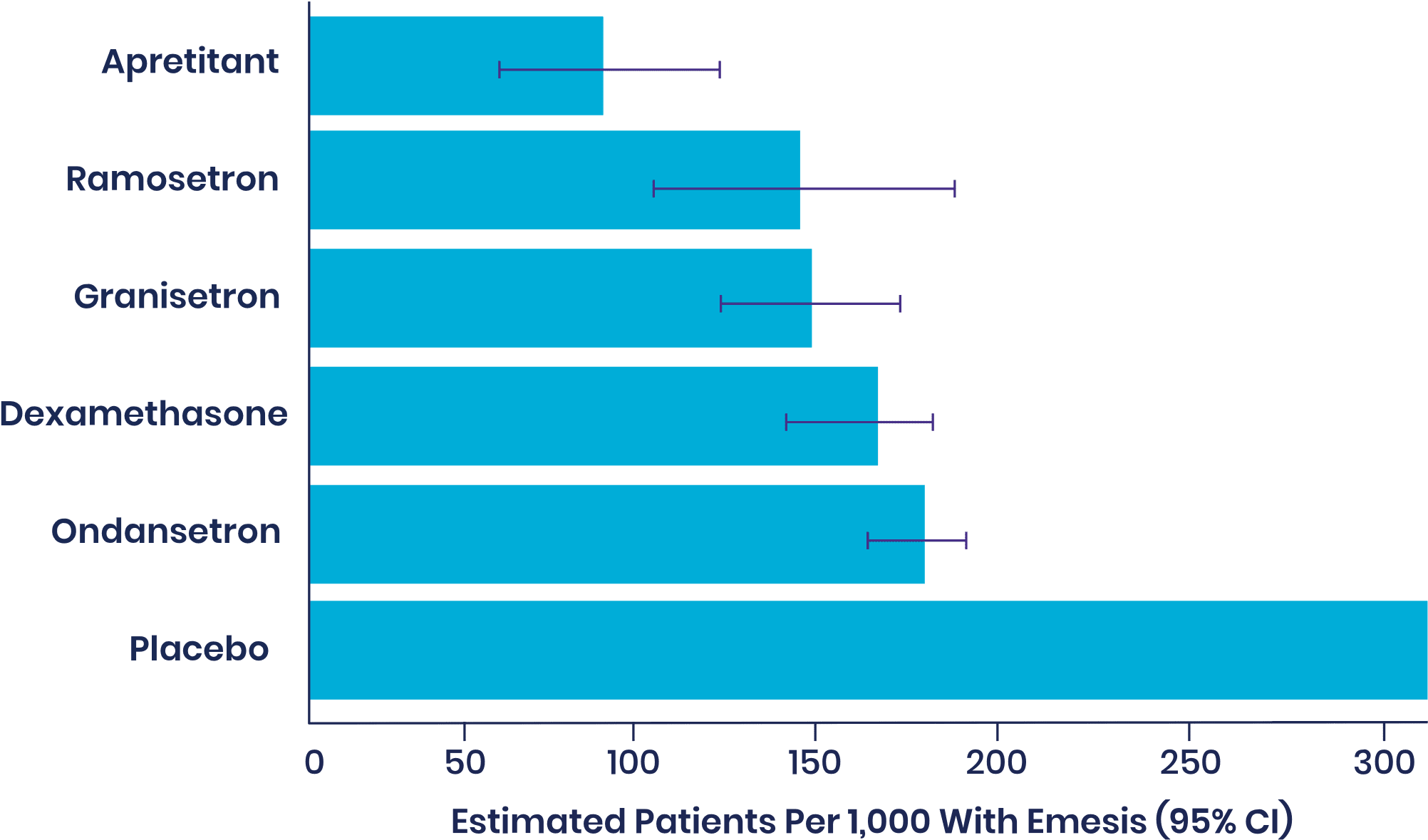
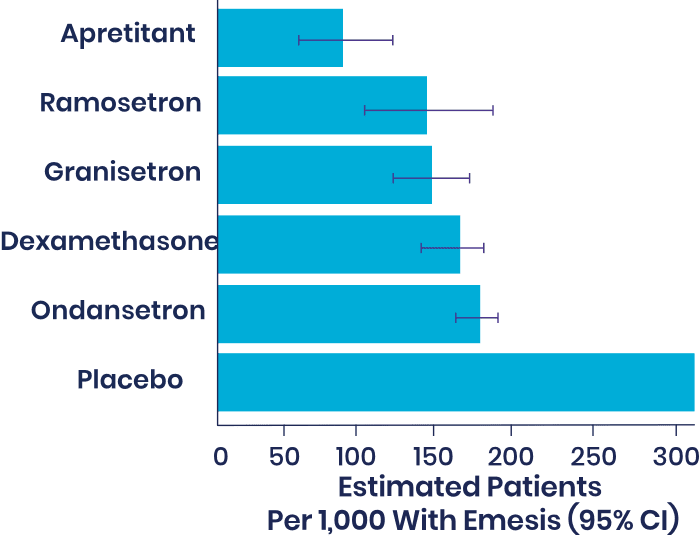
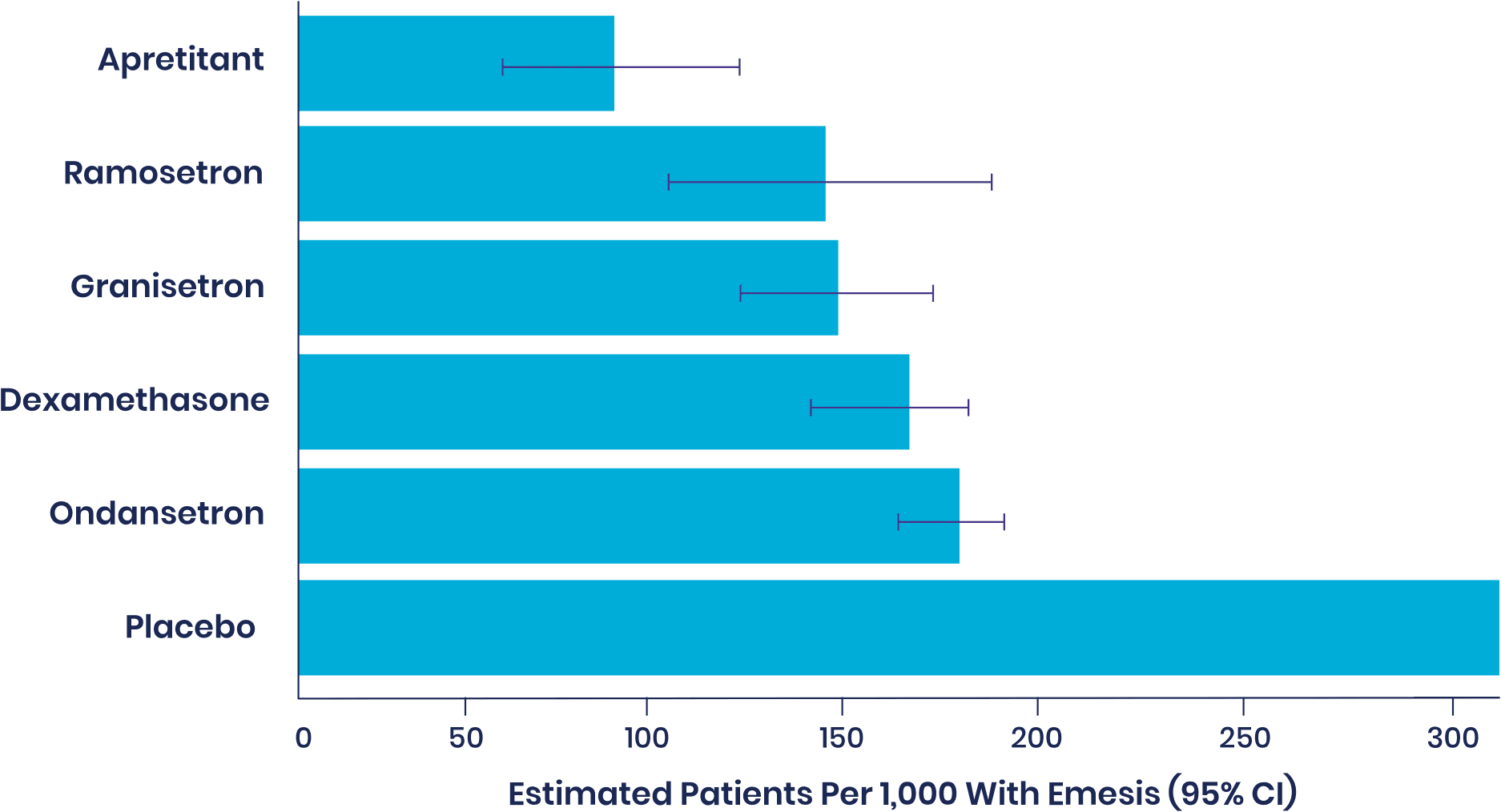
In an independent 2020 Cochrane meta-analysis of 585 studies and 97,516 patients, aprepitant was ranked as most effective single agent with high certainty of evidence among drugs with a PONV prophylactic indication, with the lowest rate of vomiting.
Ranking of most effective single-agent prophylactic with high-confidence evidence through 24 hours
Aprepitant Is Part of the Top-Performing Multimodal Approaches for the Prevention of Vomiting After Surgery1
Aprepitant-containing multimodal regimens were the most effective combinations
The most effective combination contained aprepitant
Aprepitant was included in 2 of the top 3 most effective combinations
Of 36 combination regimens analyzed in this Cochrane review (and 65 overall regimens), the top 3 most effective combinations versus placebo were:
Aprepitant / Palonosetron
Dexamethasone / Metoclopramide / Ondansetron
Aprepitant / Ramosetron
Resources
References: 1. Weibel S, Rücker G, Eberhart LH, et al. Drugs for preventing postoperative nausea and vomiting in adults after general anaesthesia: a network meta-analysis. Cochrane Database Syst Rev. 2020;10(10):CD012859. Published October 19, 2020. doi:10.1002/14651858.CD012859.
Important Safety Information
Contraindications
APONVIE is contraindicated in patients with a history of hypersensitivity to aprepitant or any component of the product, and in patients taking pimozide. Increased pimozide levels may cause serious or life-threatening reactions, such as QT prolongation.
Warnings and Precautions
Hypersensitivity Reactions: Serious hypersensitivity reactions, including anaphylaxis, during or soon after administration of aprepitant have occurred. Symptoms including dyspnea, eye swelling, flushing, pruritus, and wheezing have been reported. Monitor patients during and after administration. If hypersensitivity reactions occur, administer appropriate medical therapy. Do not administer APONVIE in patients who experienced these symptoms with previous use of aprepitant.
Clinically Significant CYP3A4 Drug Interactions: Aprepitant is a substrate, weak-to-moderate (dose-dependent) inhibitor, and an inducer of CYP3A4. Use of pimozide, a CYP3A4 substrate, with APONVIE is contraindicated. Use of APONVIE with strong CYP3A4 inhibitors (eg, ketoconazole) may increase plasma concentrations of aprepitant and result in an increased risk of adverse reactions related to APONVIE. Use of APONVIE with strong CYP3A4 inducers (eg, rifampin) may result in a reduction in aprepitant plasma concentrations and decreased efficacy of APONVIE.
Decrease in INR with Concomitant Warfarin: Use of aprepitant with warfarin, a CYP2C9 substrate, may result in a clinically
significant decrease in the International Normalized Ratio (INR) of prothrombin time.
Monitor the INR in patients on chronic warfarin therapy in the 2-week period particularly at
Risk of Reduced Efficacy of Hormonal Contraceptives: The efficacy of hormonal contraceptives may be reduced for
Use in Specific Populations
Avoid use of APONVIE in pregnant women as alcohol is an inactive ingredient in APONVIE. There is no safe level of alcohol exposure in pregnancy.
Adverse Reactions
Most common adverse reactions (incidence ≥3%) for APONVIE are constipation, fatigue, and headache and for oral aprepitant are constipation and hypotension.
Report side effects to Heron at
Indication
APONVIE (aprepitant) is a substance P/neurokinin-1 (NK1) receptor antagonist, indicated for the prevention of postoperative nausea and vomiting (PONV) in adults.
Limitations of Use: APONVIE has not been studied for treatment of established nausea and vomiting.
Please see full Prescribing Information.
Connect With Heron
How could you incorporate APONVIE as the foundation of your institution's PONV management strategy? Connect with us to find out.
Fields marked with an asterisk (*) are required.
Thank You
To report an adverse event or product complaint:
844-HERON11 (844-437-6611)
heronmc@eversana.com
For general information:
Investors and Media:
For all other inquiries, please visit the Heron Therapeutics corporate website.
Go to Heron Website